|
16 Locate the element Chlorine on your PT. Atomic mass (# of protons & neutrons) increases from left to right across a period.Atomic # (# of protons) from L to R across a period.Draw the number of shells for Periods 1 thru 7 on your PT.Write the val e-s for families I thru VIII on your PT.Elements in a period are not alike in properties.The horizontal rows of the periodic table are called periods (from repeating pattern). Li, Na, and K have similar properties, all in 1 group. Sodium (Na) has properties similar to Lithium (Li). Elementsin the same group or familyhave similar characteristics or properties.Ex.Metallic character increases from top to bottom.The vertical (up and down) columns of the periodic table (there are 18) are called groups (18)or families(I- VIII).Draw atomic structure of H & He as a class.Mass # is the # of protons PLUS the # of neutrons (each equals 1 AMU). 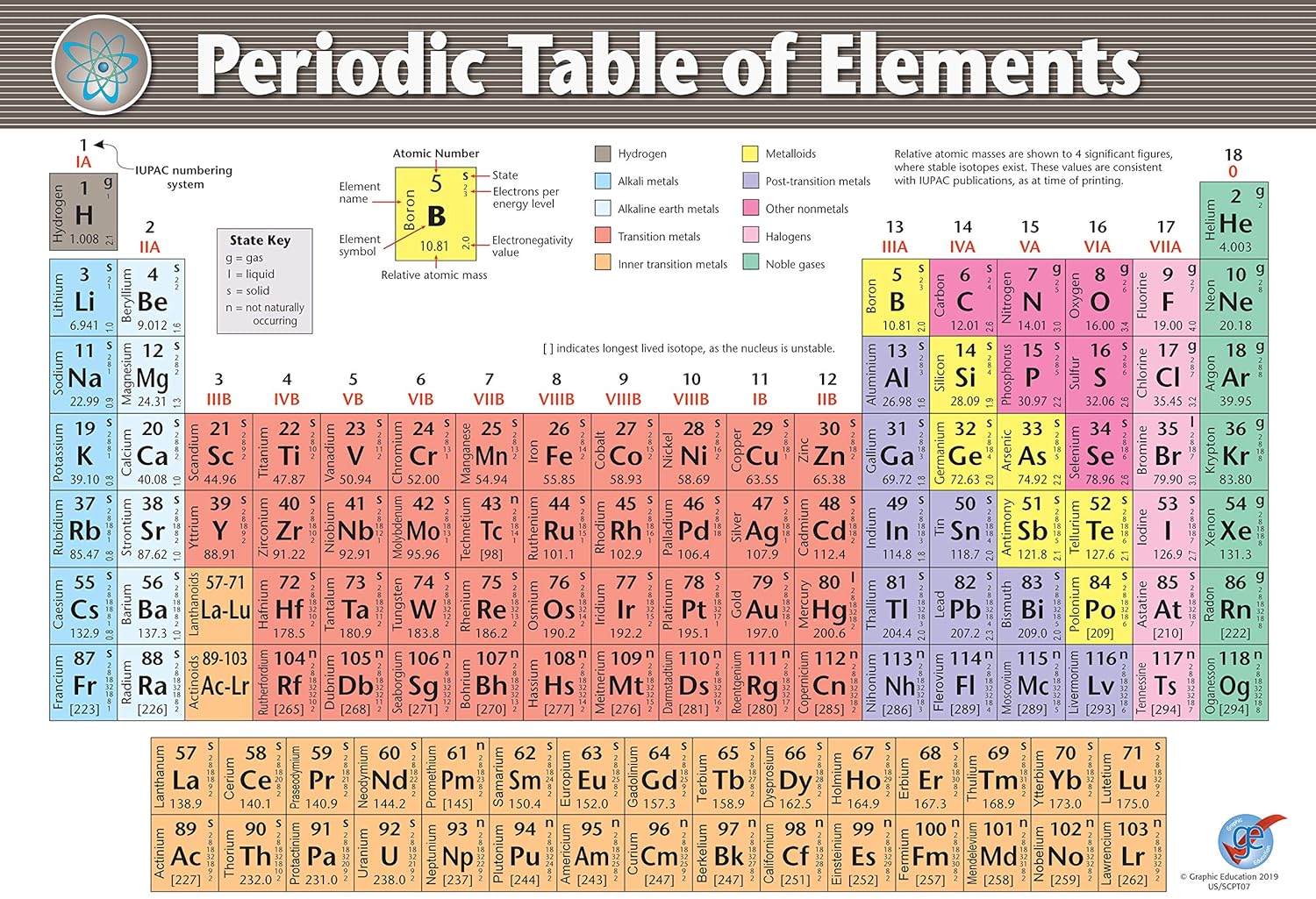
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
